X
Product Description
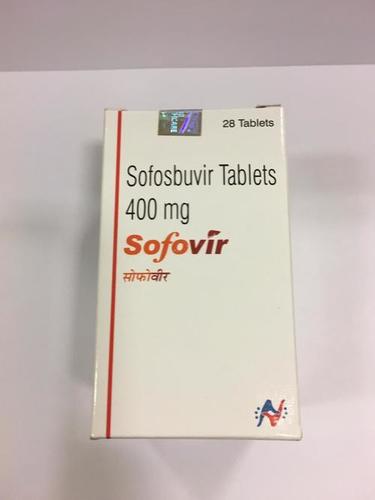
NATCO RECEIVES GENERIC SOFOSBUVIR APPROVAL FOR INDIA Hyderabad, India, March 12th, 2015 Natco Pharma Limited (NSE: NATCOPHARM; BSE: 524816) is pleased to announce that it is the 1st company in India to get approval for generic sofosbuvir tablets, 400mg, from Drugs Controller General (India). Sofosbuvir is a medicine used for chronic hepatitis C infection and sold globally by Gilead Sciences, Inc.,Natco plans to price its generic medicine at an MRP of INR 19,900 for a bottle of 28 tablets and expects to launch in India very soon. Natco had recently signed a non-exclusive licensing agreement with Gilead Sciences, to manufacture and sell generic versions of its chronic hepatitis C medicines in 91 developing countries.Enter Buying Requirement Details
Other Products in 'Sofosbuvir Tablets' category
MODERN TIMES HELPLINE PHARMA
Delhi, India
Delhi, India
5A/7, Ground Floor, Ansari Road, Darya Ganj,Delhi - 110002, India
Phone :08045803825
Direct No. : +919899980831
 |
MODERN TIMES HELP LINES PHARMA
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese